|
Since animals (including humans) breathe molecular oxygen and require it for metabolism, it is important medically. We rely on photosynthesis of plants to replenish the molecular oxygen in the atmosphere - if photosynthesis stopped, eventually the atmospheric oxygen content would drop to near zero. Molecular oxygen is very chemically reactive, and tends to form oxides by reaction with other elements and compounds quite easily. It's also essential for fossil fuel combustion. Molecular oxygen is essential for life, as it is used for respiration by many organisms. Molecular oxygen (O 2) is a diatomic molecule that is composed of two oxygen atoms held together by a covalent bond. Model of molecular or diatomic oxygen, composed of two oxygen atoms bonded together. About 99.76% of oxygen is oxygen-16.Figure 1. This is why the atomic weight of oxygen (15.9994) is so close to 16. 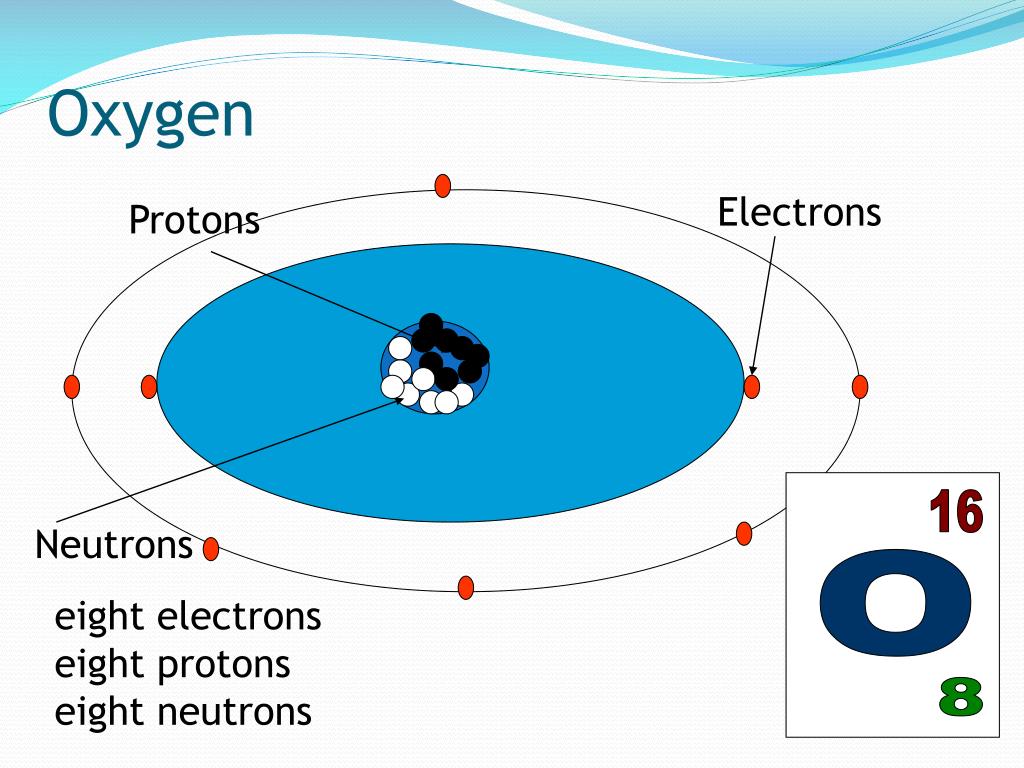
Oxygen made a good choice for the standard before much was known about isotopes because although there are 3 natural isotopes of oxygen, most of it is oxygen-16. Oxygen was the atomic weight standard for the other elements until 1961 when it was replaced by carbon 12.It's the molecule of primary importance, as far as generating bright and colorful auroras. Excited oxygen is responsible for the bright red, green, and yellow-green colors of the aurora.As stars burn hydrogen and helium, oxygen becomes more abundant. Oxygen is also the most abundant element in the Earth's crust (about 47% by mass) and the third most common element in the Universe. Although there are more hydrogen atoms in the body than oxygen atoms, they account for significantly less mass. 
This makes it the most abundant element, by mass, in the body. Much of that oxygen is part of water, H 2O.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
